What is the Difference Between Acid Strength and Acid Concentration?

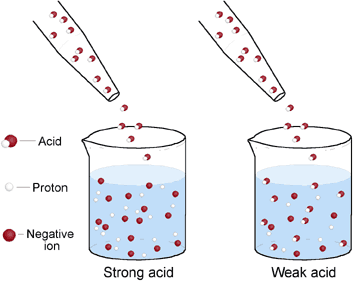
The concentration of an acid is the number of moles in the acid per liter whereas the strength of an acid is the degree of dissociation of the hydrogen ion.
* Dissociate means to split, disconnect or separate.
* Dissociate means to split, disconnect or separate.
|
Strong Acids
Hx → H+ A strong acid completely dissociates to form hydrogen ions. A strong acid is one that forms a higher concentration of H+ ions and has a PH between 1 and 3. |
Weak Acids
Hx ---> H+
<--- A weak acid dissociates partially to form hydrogen ions. A weak acid has a lower concentration of hydrogen ions and has a PH between 4 and under 7. |
This means that its possible to have concentrated weak acid solutions and dilute strong acid solutions.