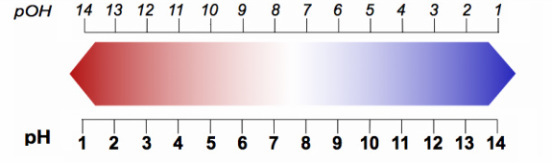
* pH + pOH must equal 14! When one is found the number can be subtracted from 14 in order to find the other!
|
[OH-]
|
[H+]
|
|
Hydroxide is a diatomic anion with a chemical formula of OH- It consists of an oxygen and a hydrogen atom held together by a covalent bond, and carries a negative electric charge. The brackets around the OH- means the concentration of it.
The mathematical formula is : [OH-]=2nd log(pOH) |
H+ ions are the measurment of hydrogen ions. The brackets around H+ mean the concentration of.
The mathematical formula is : [H+]= 2nd log[-pH] |
*[H+] x [OH-] = 1 x 10-14
Want A More Visual Example? Here is a Video on How to Do These Calculations.